ABOUT EURAP
EURAP History
EURAP is a prospective observational study of pregnancies with antiseizure medications (ASMs).
It was launched in Europe in 1999 by a consortium of independent research groups and later extended to several other nations worldwide. The aim was to collect data on the risk of antiseizure medications during pregnancy and share it in an international registry.
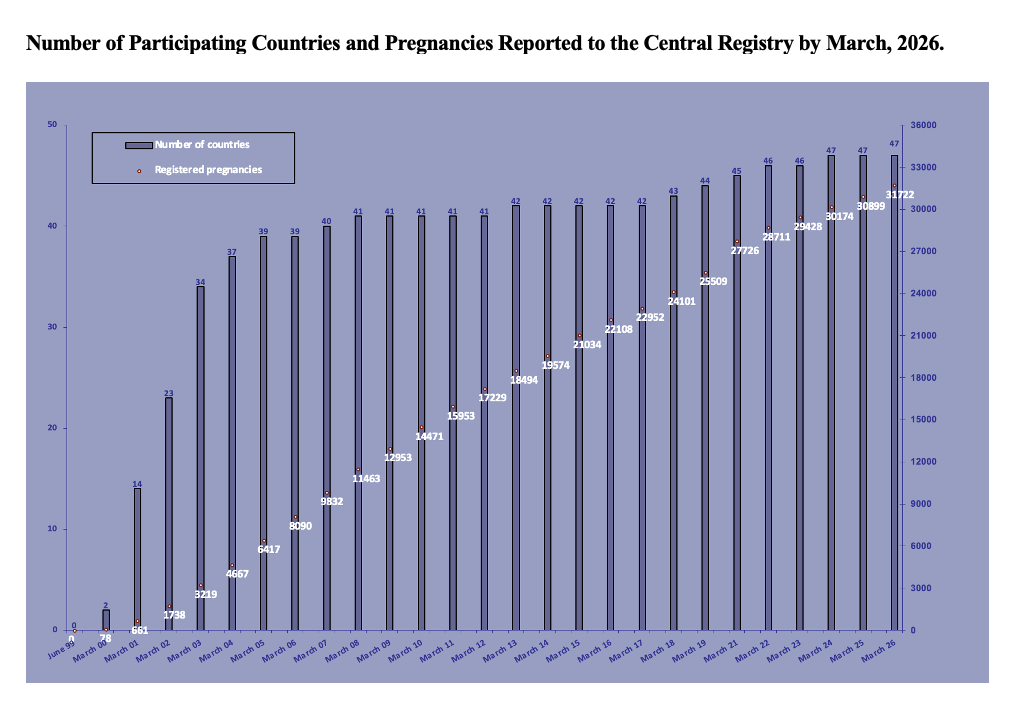
Physicians from 47 countries in Europe, Oceania, Asia, Latin America and Africa have participated to the study and more than 30,000 pregnancies have been registered so far.
Mission Statement
EURAP is an international collaboration with the vision to establish a basis for a safer treatment of epilepsy during pregnancy. The primary objective is to compare the safety of different antiseizure medications during pregnancy with respect to the risk of birth defects.
Objectives
The primary goal is to compare the risk of major congenital malformations following maternal intake of different antiseizure medications (ASMs) and their combinations. Secondary objectives include the evaluation of:
-
- any specific pattern of foetal abnormalities
- dose-effect relationships
- other risk factors
Why a pregnancy registry?
The use of ASMs during pregnancy is associated with an increased risk of birth defects.
At present, though, the information on the teratogenicity of old ASMs in humans is conflicting, due to inadequate sample size and other shortcomings of the studies completed to date. In addition, the potential harmful effects of newer ASMs are even less known. This situation prevents a rational approach to the issue of ASM treatment in women of childbearing age. To try to fill this gap, and provide meaningful conclusions with respect to the safety of different ASMs in pregnancy, it is necessary to gather data from large numbers of patients.
And this is best accomplished in an international collaboration.
How is it carried out?
EURAP is an observational study and does not interfere with the treatment prescribed by the patient’s physician. The data collected are part of the information that should be generally available during good medical care. The study does not entail any special evaluation procedure or extra visits.
The EURAP project is open to any physician or research group that can comply with the EURAP core protocol. Before starting the patient’s enrollment, physicians need to obtain approval from the Ethics Committee of their institutions.
Patient enrollment
All women taking antiseizure medications at conception are eligible for inclusion whether the indication for treatment is epilepsy or other disorders.
Recruitment is carried out by Reporting physicians after gathering two distinct informed consents from the enrolled patients, namely an informed consent for the participation to the study and another one for the data processing, as required by the EURAP core protocol.
To ensure anonymity, the patients’ personal data (date of birth or initials of their names) that may somehow lead to their identification are hidden in the Central Registry’s database and can only be seen by the Reporting physician collecting the data, in its local interface database.
To avoid selection bias, only pregnancies enrolled before foetal outcome is known and within week 16 of gestation contribute to the prospective study. Retrospective cases are also collected as they may provide valuable information, but they are not included in the comparative risk evaluation
Who can contribute?
All physicians who care for women taking antiepileptic drugs during pregnancy are invited to contribute. If interested, please go to the section ‘contact EURAP’ and complete the Form.
Funding and support
EURAP is an independent research initiative promoted and implemented by physicians working on a non-profit basis. It is administratively organised by the Central Project Commission (CPC) with members representing different geographical areas and disciplines.
The project was established and carried out under the auspices of the European Epilepsy Academy (EUREPA) and it is endorsed by the Commission on Therapeutic Strategies of the International League Against Epilepsy (ILAE).
EURAP has been supported over the years by donations from Accord Healthcare Ltd, Angelini Pharma, Betapharm Arzneimittel GmbH, Bial, DOC Generici, Ecupharma srl, Eisai Europe limited, GlaxoSmithKline, Glenmark Pharmaceuticals, GW/Jazz Pharmaceuticals, Hikma Portugal, Janssen-Cilag, Johnson & Johnson, Krka, Novartis, Pfizer, Sanofi, Teva, UCB biopharma and Zentiva. In addition, national and regional networks may receive support from the same or other pharmaceutical companies.
